About
Bio
Launched in September 2007 and written for the pharmaceutical regulatory community including regulators, academia, consultants and attorneys. Covers regulatory issues for pharmaceutical and biotech products including the areas of CMC and GMP. Specially covers the relevant issues surrounding international harmonization and helps to further the discussion about the regulatory approaches appropriate for advancing products and processes. Each issue provides an in-depth analysis of a problem area and developing regulatory initiative. Seeks to improve understanding of issues and aid the development of harmonized global regulation of pharmaceutical quality and corporate quality systems. Also reports on the key conferences and workshops where proposals for industry and regulatory guidances and initiatives are being presented and covers the US Food and Drug Administration (FDA). Includes information on international quality initiatives and harmonization efforts involving industry associations, regulatory agencies, pharmacopeias and standard-setting organizations around the globe.
Email
email@cision.one
Website
site@cision.one
Social media




Location
United States of America
Frequency
upgrade
Circulation
upgrade
Sectors
Pharmaceutical Manufacturers, Pharmaceuticals, Biotechnology
Bio
Launched in September 2007 and written for the pharmaceutical regulatory community including regulators, academia, consultants and attorneys. Covers regulatory issues for pharmaceutical and biotech products including the areas of CMC and GMP. Specially covers the relevant issues surrounding international harmonization and helps to further the discussion about the regulatory approaches appropriate for advancing products and processes. Each issue provides an in-depth analysis of a problem area and developing regulatory initiative. Seeks to improve understanding of issues and aid the development of harmonized global regulation of pharmaceutical quality and corporate quality systems. Also reports on the key conferences and workshops where proposals for industry and regulatory guidances and initiatives are being presented and covers the US Food and Drug Administration (FDA). Includes information on international quality initiatives and harmonization efforts involving industry associations, regulatory agencies, pharmacopeias and standard-setting organizations around the globe.
Website
Social media
Location
Frequency
Circulation
Sectors
Pharmaceutical Manufacturers, Pharmaceuticals, Biotechnology
Most recent articles by International Pharmaceutical Quality
Lorem ipsum dolor sit amet, consectetur adipiscing elit
Article description
Lorem ipsum dolor sit amet, consectetur adipiscing elit
Article description
Lorem ipsum dolor sit amet, consectetur adipiscing elit
Article description
Explore outlets similar to International Pharmaceutical Quality
-
SScience
Founded in 1880, with seed money from Thomas Edison, Science continues to be at the center of the very best in research across the sciences, with articles that consistently rank among the most cited in the world. Science articles are peer-reviewed, and written to appeal to both scientist and layman. Regular issue features include a news and comment section, book reviews, and coverage of scientific meetings and seminars. The News section features late-breaking news and in-depth news analysis. Compass, an opinion section, publishes letters and commentary by scientists on current science issues. The TechSight section focuses on leading edge techniques and technologies. Topics include: bioinformatics, genome sequencing, and protein sequencing. This section is published in the third issue every month. The Research section features original investigative research and reporting. Other departments include NetWatch, This Week in Science, ScienceScope, Random Samples and New Products.
ViewMMedscapeMedscape was created for medical specialists, primary care physicians, and health professionals and offers medical news, clinical trial information, review articles, journal commentary, expert columns, patient education articles and book reviews. It also includes Web-based services for verifying insurance information, Medline and other search engines, as well as a physician directory enabling users to find physicians by practice, location or specialty.
ViewSScientific AmericanEstablished in 1845, Scientific American remains the oldest continuously-published magazine in the United States. Redesigned in October 2010, the publication covers the most important and exciting research in science, health, technology, the environment, and society, exploring important ideas often months and years before other media recognizes their importance. The magazine publishes 15 foreign language editions, and regular sections include Forum, which provides a platform for external experts to comment on science policy; Science Agenda, where editors can discuss different science issues; and Insights, which profiles a scientist or researcher and analyzes their contribution to society. Other sections include Anti Gravity and Skeptic. Regular columns include The Science of Health and TechnoFiles. Publicists should feel free to pitch anytime, although they should avoid pitching individual products or new company officer announcements. They should focus on science-based stories that are better told by a journalist than an expert.
ViewTThe BMJThe BMJ is a leading international peer‑reviewed general medical journal that publishes high‑quality research, clinical reviews, expert analysis, news, opinion, education, evidence‑based guidance and commentary on clinical practice, public health, healthcare policy and related scientific and social issues affecting medicine and health professionals worldwide. It combines rigorous academic research with accessible content aimed at informing and improving healthcare decisions and outcomes globally.
ViewMMIT Technology ReviewEstablished in 1899 and written for venture capitalists and hi-tech executives as an informational source on innovative and improved technologies and their applications. Focuses on promoting the understanding of emerging technologies and their impact. Strives to promote the understanding of emerging technology and its impact on the world. Aims to promote the understanding of emerging technologies and to analyze their commercial, social and political impacts. Assists technology and business leaders including CXOs, entrepreneurs, researchers, venture capitalists and financiers as they drive the global economy.
ViewUse CisionOne to find more relevant outletsExplore journalists that write for International Pharmaceutical Quality
-
B
Contact us to find more relevant journalistsContact International Pharmaceutical Quality and get access to over 850K accurate, up-to-date media profiles.
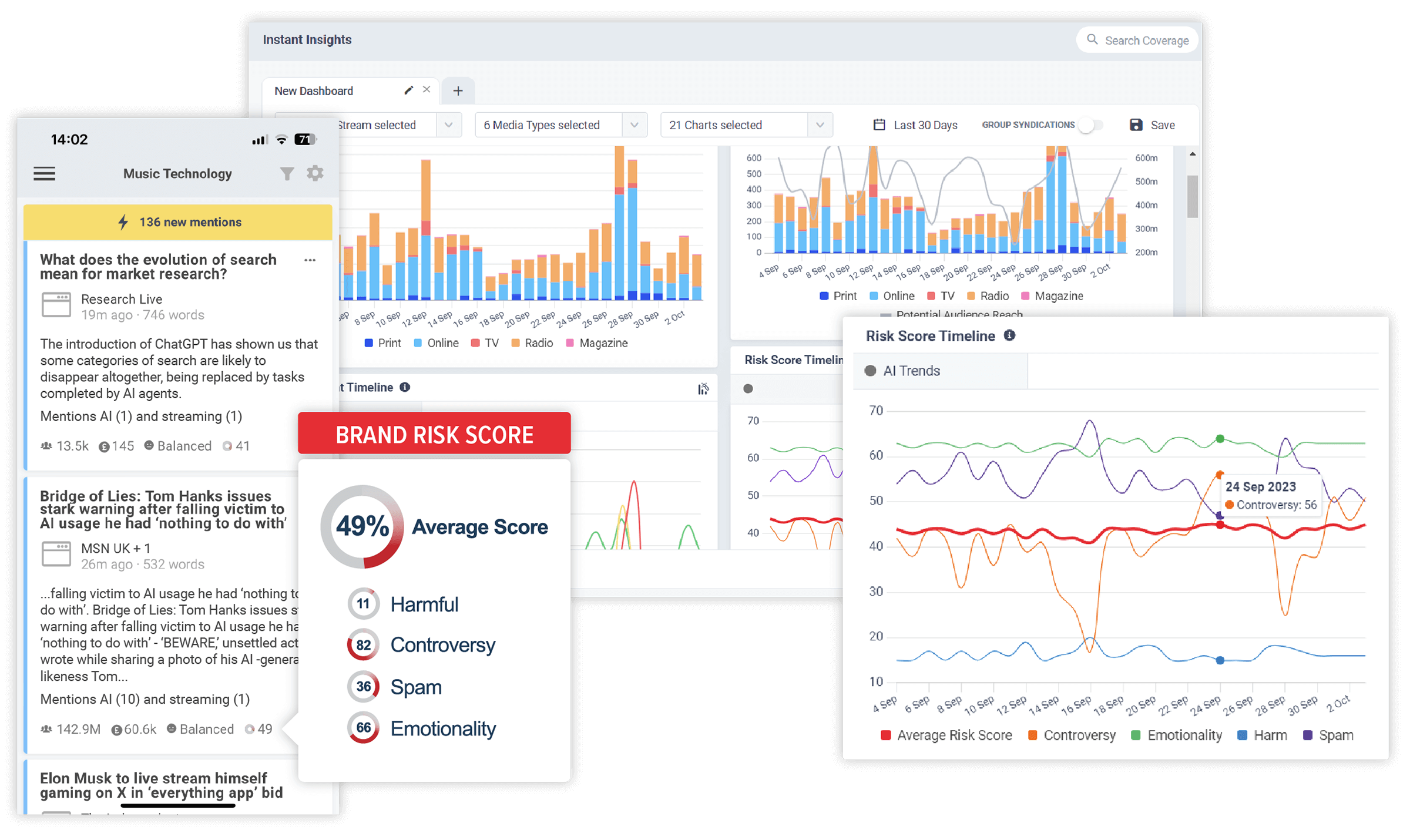
Discover the stories that impact your brand. In realtime.
CisionOne delivers relevant news, trends and conversations that matter to your brand with the world’s most comprehensive media monitoring service across Print, Online, TV, Radio, Social, Magazines, Podcasts and more.